The vitamin D-ilemma Part 3
Defining a normal, let alone an optimal, level of vitamin D is much harder than you think!
Part 3: What is a ‘normal’ vitamin D level?
In Part 1 of this vitamin D miniseries, I laid out the basics – vitamin D is not a true vitamin but a sequence of three metabolites that culminates in a secosteroid hormone called calcitriol, and is synthesised when ultraviolet B radiation from the sun hits our skin. In Part 2 I described the many ways in which this hormone influences a range of biological processes, including enhancing absorption of dietary calcium and phosphorus, combatting infection while preventing an excessive inflammatory response by the immune system, suppressing cancer growth, and maintaining healthy blood pressure.
Given calcitriol’s integral role in so many of the biological processes which maintain and restore health, it seems self-evident that being deficient in vitamin D would be inimical to health. This supposition, along with the ready availability of a blood test for vitamin D, and the relative inexpensiveness of vitamin D supplements (which can be converted into calcitriol), led to a flurry of research aimed at answering three questions: is there an optimal level of vitamin D in the human body, what dosage of vitamin D supplements is required in order to reach that level, and what effect does vitamin D supplementation have on the risk of conditions that are linked to vitamin D deficiency?
You might recall from Part 1 that when your doctor orders a blood test for ‘vitamin D’, what is actually being measured is 25-hydroxyvitamin D (also known as 25-hydroxycholecalciferol, calcifediol, and calcidiol). 25-hydroxyvitamin D is produced from either cholecalciferol (vitamin D3) generated by sun exposure or ingested in vitamin D3 supplements or vitamin D3-containing foods; or from ergocalciferol (vitamin D2). 25-hydroxyvitamin D circulates at 1000 times the concentration of calcitriol and has a half-life1 of 10-20 days, in contrast to calcitriol’s half-life of just four hours.
You might also recall from Part 1 that 25-hydroxyvitamin D is not biologically active. Only once 25-hydroxyvitamin D is converted into calcitriol, either in the kidneys or within other cells that can make it, do the biological effects of ‘vitamin D’ become manifest.
However, the level of calcitriol itself is not used as a measure of vitamin D status because of its short half-life, the fact that its formation is not directly regulated by vitamin D synthesis or intake, and because even people with severe vitamin D deficiency may have a normal or elevated calcitriol due to increased parathyroid hormone and/or activity of the 1α-hydroxylase enzyme that produces calcitriol from 25-hydroxyvitamin D.
25-hydroxyvitamin D has long been considered to be the preferred metabolite to measure in order to determine vitamin D status, on the assumption that if there is sufficient 25-hydroxyvitamin D in circulation, all the biological functions attributed to vitamin D will be optimised. Unfortunately, this assumption is overly simplistic.
Firstly, vitamin D testing is fraught with potential sources of error:
“Assessing vitamin D status by measuring serum 25(OH)D concentrations is complicated by the considerable variability of the available assays (the two most common ones involve antibodies or chromatography) used by laboratories that conduct the analyses [5,6]. As a result, a finding can be falsely low or falsely high, depending on the assay used and the laboratory.”
In fact, inter-laboratory variation in test results is as high as 15 to 20 per cent!
Secondly, roughly 85 per cent of circulating 25-hydroxyvitamin D travels through the bloodstream tightly bound to vitamin D binding protein (DBP), while around 15 per cent is more loosely bound to albumin. Only the unbound fraction (less than 1 per cent) and the albumin-bound fraction are biologically available.
The vitamin D blood test doesn’t differentiate between free and bound 25-hydroxyvitamin D. There are over 120 genetic variants of DBP, resulting in significant differences in the bioavailability of 25-hydroxyvitamin D. In postmenopausal women, free and bioavailable (albumin-bound) 25-hydroxyvitamin D concentrations were better correlated with bone density than serum total 25-hydroxyvitamin D.
There is even a case report of a woman with a genetic anomaly that resulted in a total absence of DBP. Her serum (total) 25-hydroxyvitamin D level remained too low to detect with standard hospital assays despite attempts to raise it with high-dose supplementation, but her serum calcium level was normal and she did not suffer from either rickets or osteomalacia. Apparently, she was able to maintain normal calcium metabolism with a miniscule amount of free circulating 25-hydroxyvitamin D.
And DBP-driven differences in 25-hydroxyvitamin D levels are not confined to people with rare genetic anomalies:
“Even in healthy subjects, DBP levels vary widely (tenfold), leading to corresponding variations in total 25(OH)D and 1,25(OH)2D [calcitriol], but free levels of the hormone and its precursor remain relatively constant.”
Genetic polymorphisms that are highly prevalent in people of African ancestry result in lower DBP levels in black Americans than in their white counterparts, essentially cancelling out the effect of lower total 25-hydroxyvitamin D levels in blacks:
"Community-dwelling black Americans, as compared with whites, had low levels of total 25-hydroxyvitamin D and vitamin D–binding protein, resulting in similar concentrations of estimated bioavailable 25-hydroxyvitamin D. Racial differences in the prevalence of common genetic polymorphisms provide a likely explanation for this observation... Low levels of total 25-hydroxyvitamin D probably do not indicate true vitamin D deficiency when levels of vitamin D–binding protein are also low, as in many black Americans."
Vitamin D–Binding Protein and Vitamin D Status of Black Americans and White Americans
Thirdly, there is also substantial genetic variation in the vitamin D receptor (VDR) which, when activated by calcitriol, ‘switches on’ all the biological effects attributed to vitamin D. Over 900 variants of the VDR gene have been identified; certain variants are associated with increased propensity to autoimmune diseases, type 2 diabetes, cardiovascular disease and cancer. VDR gene variance affects vitamin D metabolism, such that serum 25-hydroxyvitamin D levels may vary substantially between individuals, even at the same intake dose of vitamin D.
Interestingly, multiple genome-wide association studies conducted in Europe, United States, east Asia, and Australia have found that people who are genetically predisposed to low vitamin D, and hence have lifelong lower levels of vitamin D, do not have an increased risk of bone fractures.
In its 2011 report on dietary reference intakes for vitamin D and calcium, the US Institute of Medicine acknowledged the tenuous basis for using 25-hydroxyvitamin D levels as the indicator of vitamin D status:
“What is not clearly established is the extent to which 25OHD levels serve as a biomarker of effect. That is, there is some question as to whether levels of 25OHD relate to health outcomes via a causal pathway and can serve as predictors of such outcomes.”
In fact, since parathyroid hormone production is regulated by calcitriol that is generated in, and acts upon, cells outside of the kidneys, the best biomarker of bioavailable calcitriol – and hence of vitamin D activity – may be the serum parathyroid hormone level… although just to complicate matters even further, there are significant racial differences in the relationship between parathyroid hormone and 25-hydroxyvitamin D levels too, such that a level of only 50 nmol/L (20 ng/mL) of 25-hydroxyvitamin D maximally suppresses parathyroid hormone secretion in blacks, but not in whites or Mexican Americans.
In other words… it’s complicated!
The advances in our understanding of genetic determinants of vitamin D metabolism and activity, paired with the disappointing outcomes of vitamin D supplementation trials, has led to a very recent major shift in recommendations for vitamin D testing and supplementation. I will briefly outline the backdrop to this shift, before summarising the updated guidelines and how they were reached.
Is there an optimal level of 25-hydroxyvitamin D?
The evolutionary perspective
The pastoral Maasai and the Hadzabe hunter-gatherer peoples of Tanzania lead lives that are more similar to those of the original human beings than any other extant populations. They spend the majority of the day outdoors, wearing moderate clothing, and are exposed to daily sunshine year-round. Average serum 25-hydroxyvitamin D concentrations of Maasai were found to be 119 nmol/L (48 ng/mL), with a range of 58-167 nmol/L, and 109 nmol/L (44 ng/mL), with a range of 71-171 nmol/L for Hadzabe. Serum 25-hydroxyvitamin D concentrations were even higher in pregnant Maasai women, averaging 148 nmol/L (59 ng/mL).
Many of the recommendations issued by the most enthusiastic proponents of vitamin D supplementation, rest on the assumption that 25-hydroxyvitamin D levels like those of the Maasai and Hadzabe are the physiological norm for our species. One of these enthusiasts is Dr Michael Holick, an endocrinologist who has devoted his entire career to elucidating vitamin D’s many roles in human health. There’s no denying that Holick is a pioneering researcher, who has made outstanding contributions to the field, including isolating and identifying 25-hydroxyvitamin D and calcitriol, and developing the first clinical assays for both metabolites. (Many of the studies that I cited in Parts 1 and 2 of this miniseries were authored by Holick.) There’s also no denying that Holick has made a lot of money out of promoting vitamin D testing and supplementation. A LOT of money.
Quest Diagnostics, a major provider of pathology testing services in the US, has retained Holick as a consultant since 1979. In 2018, vitamin D tests were the fifth most common lab test covered by Medicare – largely because of the Endocrine Society’s recommendations on vitamin D testing and supplementation, published in 2011, which Holick had steered (more on that, shortly).
Holick received nearly $163,000 for consulting and other services between 2013 to 2017 from pharmaceutical companies including Sanofi-Aventis and Shire, which market vitamin D supplements; and vitamin D test manufacturers Roche Diagnostics and Quidel Corporation. Payments have diminished in recent years as Holick’s career winds down (he is now 78), but he still pocketed over $26,000 from 2017-2023 from Shire and Sanofi-Aventis.
The UV Foundation, a nonprofit arm of the now-defunct Indoor Tanning Association, which represented manufacturers of tanning beds, donated $150,000 for Dr. Holick’s research between 2004 and 2006.
Duelling ‘expert’ recommendations
Prior to the recent release of its updated vitamin D guidelines, the US Endocrine Society defined vitamin D deficiency as a level of 25-hydroxyvitamin (25 OHD) of below 50 nmol/L (20 ng/mL), vitamin D insufficiency as 50-75 nmol/L (21–29 ng/mL), and sufficiency as a 25-hydroxyvitamin D level over 75 nmol/L (30 ng/ml).
Furthermore, the Endocrine Society’s July 2011 practice guideline – on which Dr Michael Holick was the first author – speculated that young adult whites and Mexican Americans might achieve higher peak bone mass if their serum 25-hydroxyvitamin D was as high as 100 nmol/L (40 ng/mL) or even beyond. The position paper recommended testing 25-hydroxyvitamin D levels in all persons considered at risk of vitamin D deficiency, including pregnant women, breastfed infants, children, young adults, adults aged over 51 and obese people… in other words, most of the US population!
To achieve the level of 25-hydroxyvitamin D that they considered optimal for bone health, the Holick-led panel of authors suggested that infants and children may require at least 1000 International Units (IU) of supplemental vitamin D per day, while adults may need at least 1500–2000 IU per day.
But that same year – 2011 – the Institute of Medicine (IOM) published an exhaustive 1116-page review of the published evidence regarding vitamin D and calcium. They concluded that a serum 25-hydroxyvitamin D level of approximately 50 nmol/L (20 ng/mL) indicated sufficiency for nearly all of the population, and advocated Dietary Reference Intakes for vitamin D of only 600 IU per day for ages 1-70 and 800 IU per day for those aged over 70.
The IOM acknowledged that there were significant information gaps and called for further research to help identify the optimal level of vitamin D for both bone health and nonskeletal outcomes, in different age and racial groups, and to clarify whether serum 25-hydroxyvitamin D is an appropriate “biomarker of effect”.
Unsurprisingly, Dr Michael Holick strongly advocated for his group’s recommendations and criticised the IOM’s methods for calculating the threshold for vitamin D deficiency. And many commercial pathology labs, including Quest and LabCorp, adopted the higher Endocrine Society cutpoint for diagnosing vitamin D deficiency that Holick endorsed, rather than the lower IOM standard. As a result, testing for vitamin D skyrocketed, and so did the number of people taking vitamin D supplements.
Just as the IOM authors had desired, the ensuing 13 years saw a flurry of vitamin D research. Many of those studies were randomised controlled trials, which sought to clarify whether the association between low 25-hydroxyvitamin D and multiple diseases ranging from bone fractures to schizophrenia, dementia and fatal cancer, documented in many prior studies, was in fact causal.
VITAL failure
The largest of those trials was the VITamin D and OmegA-3 TriaL (VITAL), which tested the effects of taking 2000 IU of supplemental vitamin D per day versus placebo, for five years, in almost 26,000 adults over age 50. The trial’s primary outcomes were prevention of cancer and cardiovascular disease, but data were collected on many other disorders, including diabetes, hypertension, cognitive decline, depression, respiratory disorders, and autoimmune diseases.
Disappointingly, there was no reduction of cancer incidence, major cardiovascular events, other cardiovascular endpoints, or all-cause mortality among VITAL participants who took 2000 IU of vitamin D per day, although there was a trend towards improved cancer survival.
There was also no reduction in fracture risk in “generally healthy midlife and older adults who were not selected for vitamin D deficiency, low bone mass, or osteoporosis”. It’s important to point out that participants’ average baseline 25-hydroxyvitamin D level was 75 nmol/L, with those who received 2000 IU of vitamin D reaching 102 nmol/L (41 ng/mL) after two years. There were too few participants with very low 25-hydroxyvitamin D levels (below 30 nmol/L [12 ng/mL]) to confidently detect any benefit of supplementation in this subgroup of people.
There was one very promising outcome from VITAL: a daily dose of 2000 IU of vitamin D for five years, with or without omega 3 fatty acids, reduced the risk of new onset autoimmune disease by 22 per cent.
Another study, the Vitamin D and Type 2 Diabetes (D2d) trial, sought to test the effects of high dose vitamin D supplementation on diabetics and prediabetics. D2d also yielded mostly disappointing results. People with prediabetes who took 4000 IU of vitamin D per day did not have a significantly lower risk of being diagnosed with diabetes after a median follow-up of 2.5 years, than those taking placebo. Type 2 diabetics who took 4000 IU of vitamin D3 daily for 48 weeks did not have any improvement in their lipid profile, C-reactive protein or cardiovascular risk (calculated according the American College of Cardiology/American Heart Association [ACC/AHA] guidelines), although those who were not taking any cholesterol-lowering medication did have any improvement in triglyceride levels.
But there was some good news: in a metanalysis of randomised controlled trials, people with prediabetes whose serum 25-hydroxyvitamin D level was at least 125 nmol/L (≥50 ng/mL) after high-dose vitamin D3 (cholecalciferol) supplementation, the risk of developing diabetes was reduced by 76 per cent, with a three-year absolute risk reduction of 18.1 per cent.
The 2024 Endocrine Society vitamin D guidelines
The disappointing results of VITAL and many other intervention trials prompted a major revision of the Endocrine Society’s practice guidelines, which was published in June.
The evidence review undertaken by the panel that developed the guidelines found:
Low-certainty evidence that vitamin D supplementation may reduce respiratory tract infections in children and adolescents;
No significant effect of supplementation on cardiovascular events (high certainty), total cancer (high certainty), mortality (high certainty), fracture (high certainty), stroke (moderate certainty), or kidney stones/kidney disease (low certainty) in healthy adults aged 19 to 74 years;
A very small reduction in mortality among adults older than 75 years with high certainty of evidence;
Low-certainty evidence of possible benefit of vitamin D supplementation in pregnant women on various maternal, fetal, and neonatal outcomes;
Moderate certainty of evidence that vitamin D supplementation reduces progression to diabetes in adults with prediabetes;
High-dose intermittent vitamin D may increase falls, compared to lower-dose daily dosing.
The laundry list of groups considered ‘at risk’ of vitamin D deficiency in their 2011 document was pared right back, with only children under 18, pregnant women, adults aged over 75 and adults with prediabetes, identified as groups who might benefit from vitamin D supplementation. Recommended dosages are 600 IU per day for those aged 1-70 years and 800-900 IU per day for those aged over 70 years, although the authors noted that clinical trials for prevention of respiratory infections in children used an average of 1200 IU per day, and those for prevention of pregnancy complications used an average of 2500 IU per day.
The practice guidelines explicitly recommend against testing for blood vitamin D levels in the general population, including those with darker skin or obesity, because 25-hydroxyvitamin D levels that provide outcome-specific benefits have not been established in clinical trials.
In adults aged 50-74, even those whose baseline 25-hydroxyvitamin D level was in the range considered insufficient or deficient – less than 50-60 nmol/L (20-24 ng/mL) – did not have a clear net benefit from vitamin D supplementation; there was also no significant difference in mortality risk in studies that found a benefit of supplementation in participants aged 75 years or older, between participants with baseline 25-hydroxyvitamin D level less than 50 nmol/L (20 ng/mL) and those whose level was considered sufficient:
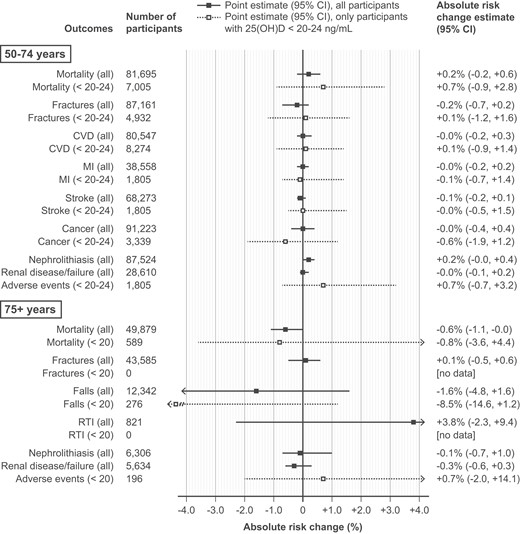
The guideline acknowledged that many important questions about vitamin D still cannot be answered. The optimal vitamin D intake and 25-hydroxyvitamin D level for disease prevention is still uncertain. And it is as yet not known whether vitamin D supplementation can prevent certain diseases in which an association with low 25-hydroxyvitamin D has been documented, because randomised clinical trials either have not been performed or are still in progress. There was also a disconcerting lack of evidence on whether the benefits of vitamin D supplementation varied based on skin pigmentation:
“The panel discovered that no randomized clinical trials have directly assessed vitamin D related patient-important outcomes based on participants’ skin pigmentation, although race and ethnicity often served as presumed proxies for skin pigmentation in the literature.”
Navigating Complexities: Vitamin D, Skin Pigmentation, and Race
In an article accompanying the updated clinical practice guidelines, titled ‘Vitamin D Insufficiency and Epistemic Humility: An Endocrine Society Guideline Communication’, the Endocrine Society acknowledged that the definitions of vitamin D deficiency and insufficiency contained in its 2011 guidelines have not been borne out by the results of subsequent randomised controlled trials, and that ‘epistemic humility’ – the willingness to admit that you’ve been wrong, and that you don’t know everything – compels them to abandon these definitions and to “advocate for additional research to assess whether discrete 25(OH)D thresholds will specifically predict important net benefit with vitamin D supplementation.”
Naturally, not every vitamin D researcher agrees with the new Endocrine Society guidelines! For example, an article published in January 2024 argued for a daily dose of 2000 IU of vitamin D across the adult population, contending that observational trials had documented the association of higher 25-hydroxyvitamin D levels with lower mortality and risk of multiple diseases, while clinical trials had demonstrated the safety of the comparatively high-dose supplementation required to achieve such 25-hydroxyvitamin D levels.
… except that high levels of 25-hydroxyvitamin D, and daily dosages of 2000 IU or higher of vitamin D, have been found to be associated with adverse health outcomes in black Americans.
And then there's the fact that multiple studies have found a U-shaped or reverse J-shaped relationship between serum 25-hydroxyvitamin D levels and adverse health outcomes - that is, both low and high levels are hazardous.
An observational cohort study (the Copenhagen vitamin D study [CopD] ) found that a 25-hydroxyvitamin D level of 70 nmol/L (28 ng/mL) was associated with the lowest cardiovascular disease mortality risk, with both lower and higher levels of 25-hydroxyvitamin D associated with heightened risk. (Note that women appear to have a wider tolerance range for 25-hydroxyvitamin D levels than men, with respect to cardiovascular mortality risk.)
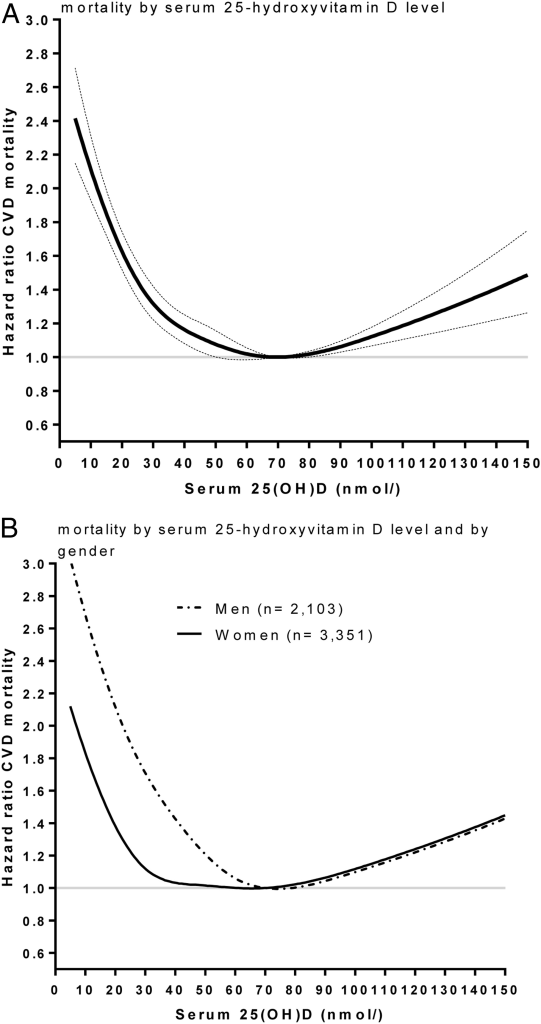
The same CopD study found that the sweet spot for all-cause mortality was considerably lower, at 50-60 nmol/L (20-24 ng/mL), in both the entire population sample and in those aged 60 and over:
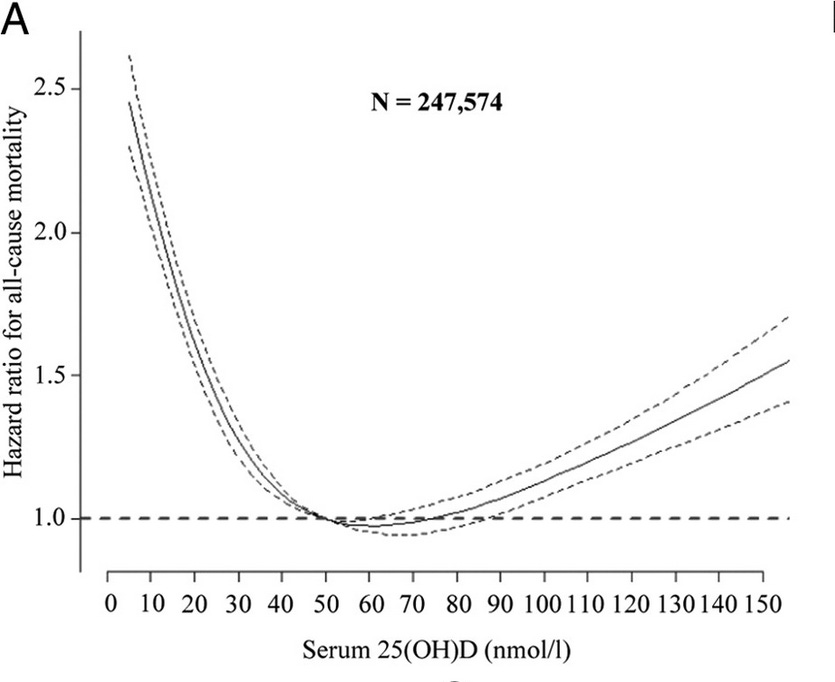
A meta-analysis of 14 prospective cohort studies based in the United States, Japan, New Zealand and Europe, pitched that sweet spot somewhat higher, at 75–87.5 nmol/L. The reason for the difference is not clear, but may be accounted for by greater genetic variation in vitamin D binding protein and vitamin D receptor activity in ethnically diverse populations, as discussed in Part 3.
Yowsers!!! How is anyone supposed to make sense of this hot mess?
One possible reason why randomised controlled trials of vitamin D supplementation have failed to live up to the promise of the observational studies which found an association between low 25-hydroxyvitamin D levels and various diseases, is reverse causation – that is, the disease process causes the (apparently) low vitamin D level, rather than the other way around. I’ll delve into the evidence for the reverse causation hypothesis in the next instalment of this miniseries.
As always, I look forward to reading your thoughts and reflections:
For information on my private practice, please visit Empower Total Health. I am a Certified Lifestyle Medicine Practitioner, with an ND, GDCouns, BHSc(Hons) and Fellowship of the Australasian Society of Lifestyle Medicine.
And finally, this post has taken me approximately 30 hours to research and write. The breadth, depth and complexity of the vitamin D research literature is truly mind-bending. I make all my posts freely available to all readers, because I believe we all deserve access to information that helps us to take greater control over our health. But I rely on my small core of paid subscribers to provide this service to those who genuinely can’t afford it.
Half-life refers to the time required for the concentration of a substance to be reduced by half.





my impression of the situation regarding testing and supplementation is that it is probably the modern version of snake oil. In fact Dr T Colin Campbell, who should know a thing or 2 on this subject, seems to pretty much dismiss nearly all attempts to make pronouncements on the how the nutritional components of food actually work, what is adequate and if supplements are even needed. When asked in a video around about 2020, what he eats, he just said "what my wife cooks" It can be assumed he meant she only provided WFPB meals, but he seems to avoid most attempts at reductionist analysis.
Thanks again for these in depth articles on Vitamin D.
Most of what I see about Vitamin D is people claiming it is a wonder supplement. However, it does have some detractors like this recent article basically saying Vitamin D promotes cancer and that we should not supplement it.
https://jimstephensonjr.substack.com/p/the-progression-of-stupidity?
Lower levels of vitamin D may reduce prostate cancer risk in older men. By contrast, levels of vitamin D did not predict incidence of colorectal or lung cancers.”
https://www.sciencedirect.com/science/article/pii/S2211383518303526
https://www.nature.com/articles/boneres201723
https://journals.plos.org/plosone/article
Now let’s look at a cancer that is typically 100% fatal, Pancreatic Cancer.
“Higher vitamin D concentrations were associated with a 3-fold increased risk for pancreatic cancer (highest versus lowest quintile, >65.5 versus <32.0 nmol/L: OR, 2.92; 95% CI, 1.56-5.48, Ptrend = 0.001) that remained after excluding cases diagnosed early during follow-up.”
https://aacrjournals.org/cancerres/article/66/20/10213/526311/A-Prospective-Nested-Case-Control-Study-of-Vitamin